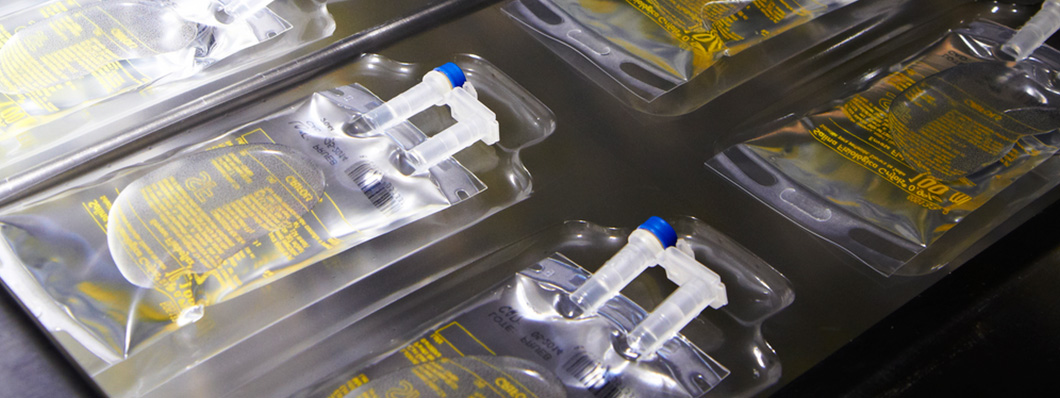
Bolsas para soluciones parenterales
Innovación tecnológica y nuevos materiales
Ficha técnica
Tipo de innovación: Aparato; Sistema; Procedimiento
Ámbito: Farmacia hospitalaria
Líder de innovación: Grifols i Lucas, Víctor
Año: 1989
Etapa: 1972-2002
Difusión: Internacional
Impacto económico: Alto
Nivel de innovación: Disruptivo
Patente: Sí
Relaciones interdisciplinares: -
Grifols comenzó a fabricar soluciones parenterales para el sector de la farmacia hospitalaria en los años 50. Estos medicamentos requieren los niveles de seguridad más elevados, ya que se inyectan directamente en el torrente sanguíneo sin ninguna barrera biológica de por medio. En consecuencia, es preciso seguir procesos extremadamente estrictos para su manipulación y, en general, garantizar la ausencia de bacterias, virus y otros microorganismos.
Además de dedicarse a la fabricación de soluciones parenterales, Grifols tenía experiencia en el desarrollo de envases y en la mejora de los procesos de llenado y esterilización de estos medicamentos. Asimismo, Grifols era capaz de adaptarse rápidamente a las innovaciones tecnológicas del sector y aumentar así la seguridad y fiabilidad de sus productos.
Del vidrio al plástico
Las soluciones parenterales destinadas a la farmacia hospitalaria se solían envasar en recipientes de vidrio, puesto que se trataba de un material neutro adecuado para el transporte y la conservación de medicamentos administrados por infusión intravenosa. Sin embargo, a mediados de los años 80, una serie de estudios llevados a cabo con el propósito de identificar una alternativa más flexible a los envases de vidrio convencionales concluyeron que el PVC era el mejor material para conservar dichas soluciones. El PVC también era la opción preferida del personal de enfermería, ya que con él se eliminaba el problema de las roturas de las bolsas.
Para responder a esta tendencia de mercado, Grifols optó por introducir envases flexibles para sus productos de fluidoterapia. No obstante, era preciso resolver primero algunos problemas para garantizar la seguridad y funcionalidad de los nuevos envases y adaptar el proceso de llenado al nuevo material.
La doble bolsa
Si bien en un principio se creía que el plástico era impermeable, no tardó en comprobarse lo contrario: parte del líquido que se aloja en un contenedor plástico acaba por evaporarse en cuestión de tiempo. Esto planteaba problemas en los hospitales cuando se precisaba infundir una cantidad específica de solución y al utilizar el producto, el volumen contenido en el envase era inferior al esperado. La mayoría de las empresas respondió a este problema introduciendo la bolsa de solución intravenosa en una segunda bolsa para evitar la evaporación. La doble bolsa estaba diseñada para garantizar mayor protección y seguridad de dos maneras: por un lado, reducía el riesgo de contaminación derivado del manejo; y, por otro, la bolsa adicional impedía la evaporación del contenido líquido.
“El paso del vidrio al plástico obligó a modificar los procesos industriales de llenado y esterilización de soluciones parenterales. Víctor Grifols i Lucas, director técnico de Grifols en aquella época, resolvió el problema diseñando una máquina que se patentó en 1990.”
Un nuevo sistema de llenado
El paso del vidrio al plástico obligó a modificar los procesos industriales de llenado y esterilización de soluciones parenterales. Víctor Grifols i Lucas, director técnico de Grifols en aquella época, resolvió el problema diseñando una máquina que se patentó en 1990. Con el nuevo procedimiento, la bolsa vacía se colocaba en un molde y a continuación se hacía el vacío. A continuación, la bolsa se conectaba a un depósito con la solución, que se dispensaba a presión atmosférica. Una vez llena la bolsa, se sellaba el orificio de entrada. Con este sistema, la solución entraba en la bolsa directamente, sin válvulas o juntas intermedias, lo que reducía el riesgo de contaminación por partículas.
El uso de la presión interna para garantizar la esterilidad
La única forma de garantizar que una solución está libre de pirógenos consiste en fabricarla en unas condiciones totalmente estériles. Para lograrlo, suele ser necesario utilizar autoclaves en las que la solución se somete a tratamiento térmico a altas presiones durante varias horas. El paso del vidrio al plástico planteó un problema, ya que algunas bolsas se rompían debido al descenso brusco de presión y temperatura durante la etapa de enfriamiento y descompresión. Grifols resolvió este problema analizando las presiones internas que se registraban en la autoclave y aplicando contrapresiones mucho más graduales, lo que reducía considerablemente el esfuerzo mecánico al que se veían sometidas las bolsas en la etapa de enfriamiento.
Puertos de inyección e infusión integrados
Otra de las innovaciones que introdujo Grifols fue integrar el puerto de inyección y el de infusión en una misma pieza. Hasta entonces, estos puertos eran independientes y constaban de varios componentes. El puerto de inyección e infusión integrado de Grifols ofrecía diversas ventajas, en relación tanto con el proceso de fabricación como con la seguridad y la ergonomía. Así, el puerto integrado era más fácil de sujetar y más rígido, lo que reducía el riesgo de pinchazos accidentales. Además, al carecer de piezas rompibles que desechar, el nuevo diseño era también más respetuoso con el medio ambiente.
Envases plásticos para soluciones parenterales
Flebobag®: la bolsa de PVC flexible de Grifols
El primer envase plástico de Grifols para soluciones parenterales fue la bolsa Flebobag®, cuyo éxito obligó a Grifols a recurrir a dos proveedores de bolsas de PVC a fin de satisfacer la demanda del producto. En 2003, la empresa producía al año 14 millones de bolsas de soluciones intravenosas, que se destinaban principalmente a la red hospitalaria española.
Fleboflex®: bolsas de polipropileno y fabricación automática
En 2005, Grifols comercializó la bolsa Fleboflex®, fabricada con polipropileno. La creciente demanda del producto en los años siguientes propició en 2009 la automatización de las líneas de producción y la construcción de una nueva fábrica de soluciones parenterales en envase flexible de polipropileno. Con las nuevas instalaciones, aumentó la capacidad en 30 millones de bolsas por año hasta alcanzar un total de 44 millones.
Bibliografía
Grifols i Lucas, V. (1960). Perfeccionamiento en los medios de control automático de la presión y tiempo de esterilización en autoclave. (Oficina Española de Patentes y Marcas Patente Nº 261354). Enlace
Grifols i Lucas, V. (1966). Bolsa para la transfusión de soluciones gota a gota dotada de racord de alimentación con cierre estanco que se abre desde el exterior. (Oficina Española de Patentes y Marcas Patente Nº 120126). Enlace
Grifols i Lucas, V. (1989). Procedimiento perfeccionado para el llenado de bolsas con líquidos a perfundir e instalación para la puesta en práctica de dicho procedimiento. (Oficina Española de Patentes y Marcas Patente Nº ES2016489 (A6)). Enlace
Grifols i Lucas, V. (1989). Procedimiento perfeccionado para recubrir una bolsa de plástico que contiene líquido para perfusión con otra bolsa, máquina para la puesta en práctica de dicho procedimiento y bolsa obtenida en el mismo. (Oficina Española de Patentes y Marcas Patente Nº ES2016492 (A6)). Enlace
Roura Fernández, C. y Menéndez Cortina, J. (2005). Bolsa para soluciones terapéuticas. (Oficina Española de Patentes y Marcas Patente Nº 200501664). Enlace
Grifols, S.A. (2011). ¿Y si lo hacemos así? Patentes de Víctor Grífols i Lucas. Barcelona: Grifols, S.A.
Roura Fernández, C., Garcia Garcia, J.A., Llorens Masas, E. y Marzo Adam, N. (2016). Double bag. Contenedor para una solución de proteínas plasmáticas humanas y procedimiento de preparación del mismo. (Oficina Española de Patentes y Marcas Patente Nº 201631198). Enlace
Innovaciones relacionadas
FLUIDOTERAPIA
Soluciones intravenosas